Corrosion can be simply defined as a gradual degradation of a metal surface. It is a natural process that occurs as the result of the chemical and/or electrochemical reaction the metal has with its environment. Unfortunately, it is inevitable; it is only the rate of corrosion than can be altered by various processing and/or finishing methods.
Some Common Causes of Corrosion in Hydraulic & Automotive Systems
Corrosion vs. Oxidation – Are They the Same?
Because rust can be a manifestation of both of these processes, they are often thought to be the same. They are not, because the source of the rust is different.
- Oxidation in hydraulic systems occurs when the oxygen surrounding the system comes into contact with the water trapped in the fluid lines.
- Corrosion is the result of moisture and wet weather conditions—rain, snow or sleet.
This is why corrosion and/or oxidation are inevitable; there will never be a condition in which there is no moisture or oxygen in the environment.
However, premature corrosion of parts is usually a result of production methods, welding and environmental conditions.
How Corrosion Begins
Virtually any machining, mechanical cutting, welding or fabrication process will degrade surface conditions and properties by enabling the accumulation of surface contaminants, such as grease, dirt, iron and other metallic particles. Stainless steel has a naturally occurring oxide layer that is corrosion resistant. However, the formation of this layer can be disrupted by the imbedding of these contaminants. When and where this layer is disrupted, corrosion can begin.
Types of Corrosion
While it is easy to distinguish the concept of general corrosion from oxidation, there are actually various types of corrosion:
- Pitting corrosion, or pitting, is a form of extremely localized corrosion by which small holes are made in a material. It is more difficult to detect and predict and, therefore, more dangerous to the material than uniform corrosion.
- Crevice corrosion occurs when a stagnant solution or fluid in a crevice attacks a metal surface. It can form under gaskets, washers, fastener heads, threads and in other micro-environments.
- Galvanic corrosion occurs between two different metals, when they are in electrical contact and exposed to an electrolyte.
- Stress corrosion cracking (SCC) is the result of a combination of stress on the metal and a corrosive environment, leading to the formation (and expansion, over time) of a crack.
- Microbiologically influenced corrosion (MIC), also known as microbial corrosion and biological corrosion, is corrosion caused by bacteria.
Metal Finishing Methods to Prevent Corrosion
A finishing method that removes the surface metal and its contaminants is the solution to premature corrosion. There are a number of finishing methods available, and each has its pros and cons.
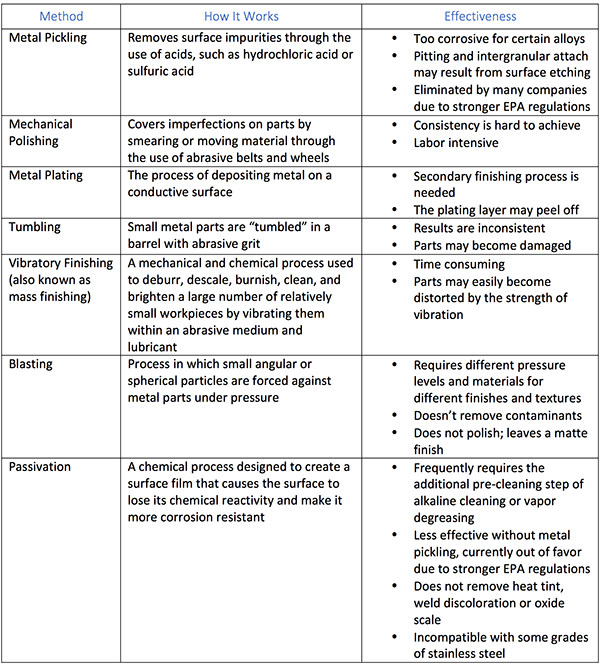
What is Electropolishing, and What Are Its Benefits to the Automotive Industry?
Oil retention is critical in any application that requires friction, such as hydraulics. If the finish of a part is too smooth, it cannot effectively hold onto the lubricant. If it is too rough, then there will be too much friction, and the part will wear down prematurely.
Certain finishing methods, such as grinding, honing and mechanical polishing are necessary in fabricating and shaping parts for use in such systems. However, they all leave surface contaminants, such as debris, dust particles and dirt that can become a source or trigger for corrosion. Additionally, they can embed such contaminants within the metal itself, so that standard cleaning and polishing methods are ineffective at removing them.
While the processes described in the table can be effective in certain applications, electropolishing offers features that make it more universally beneficial.
Because the part is immersed in a chemical bath:
- Both the surface contaminants, as well as those embedded in a part can be removed.
- There is no risk of damage or distortion, as there is with tumbling or vibrating.
- All areas of a part are reached and cleaned equally, from the tiniest of threads to the most complex configurations.
Electropolishing can also be consistently and tightly controlled, holding tolerances of +/-.0001”, and it will not change thermal properties, case hardness or cause a heat-treated alloy to warp.
If you are serious about reducing corrosion and improving function, it is an option well worth investigating.
Scott Potter is the vice president of sales at Able Electropolishing Co., Inc., the world’s largest electropolishing specialist. Scott manages sales and marketing efforts, which largely include educating metalworking companies about the effectiveness and added value of electropolishing and other types of metal finishing they provide.














